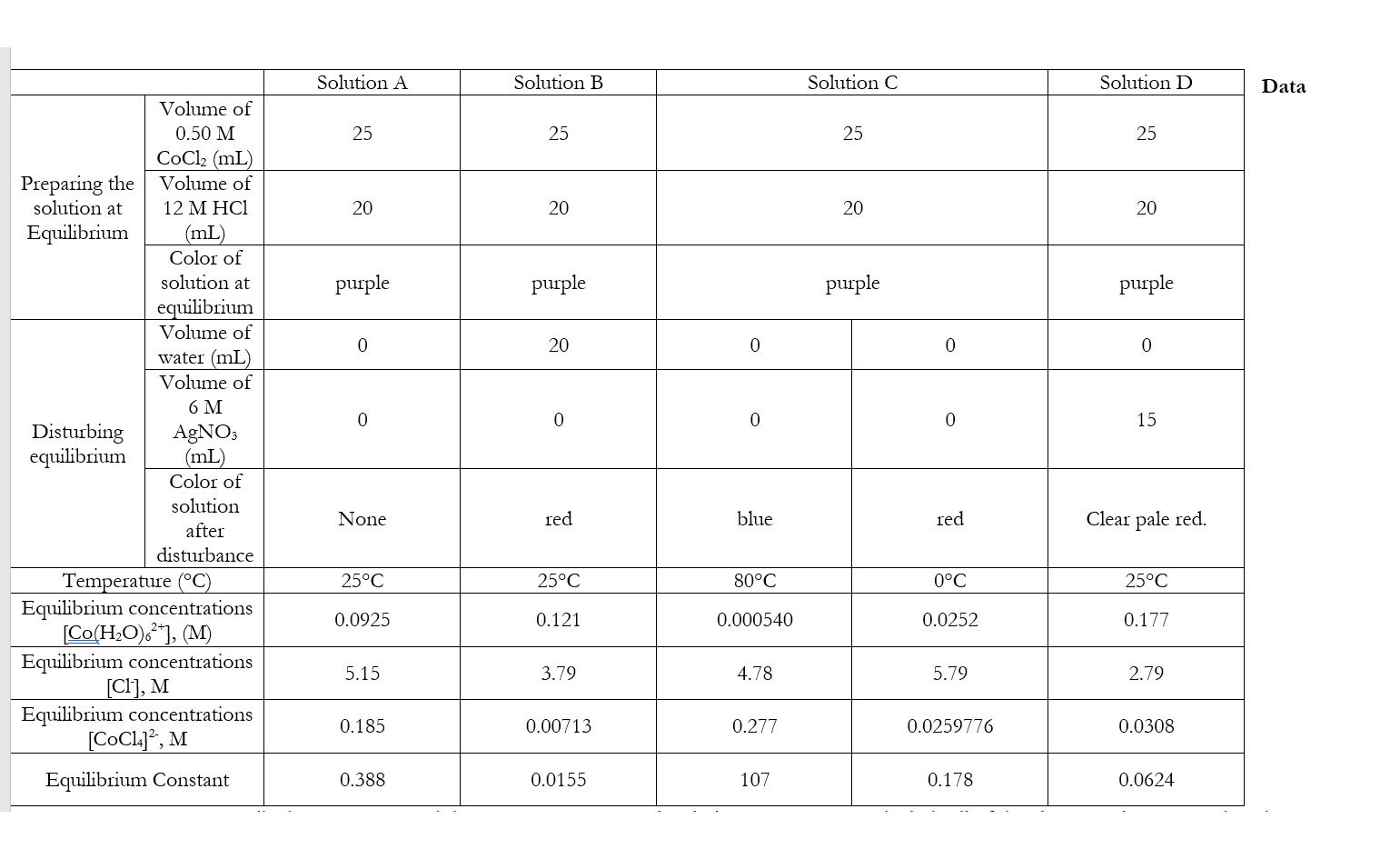
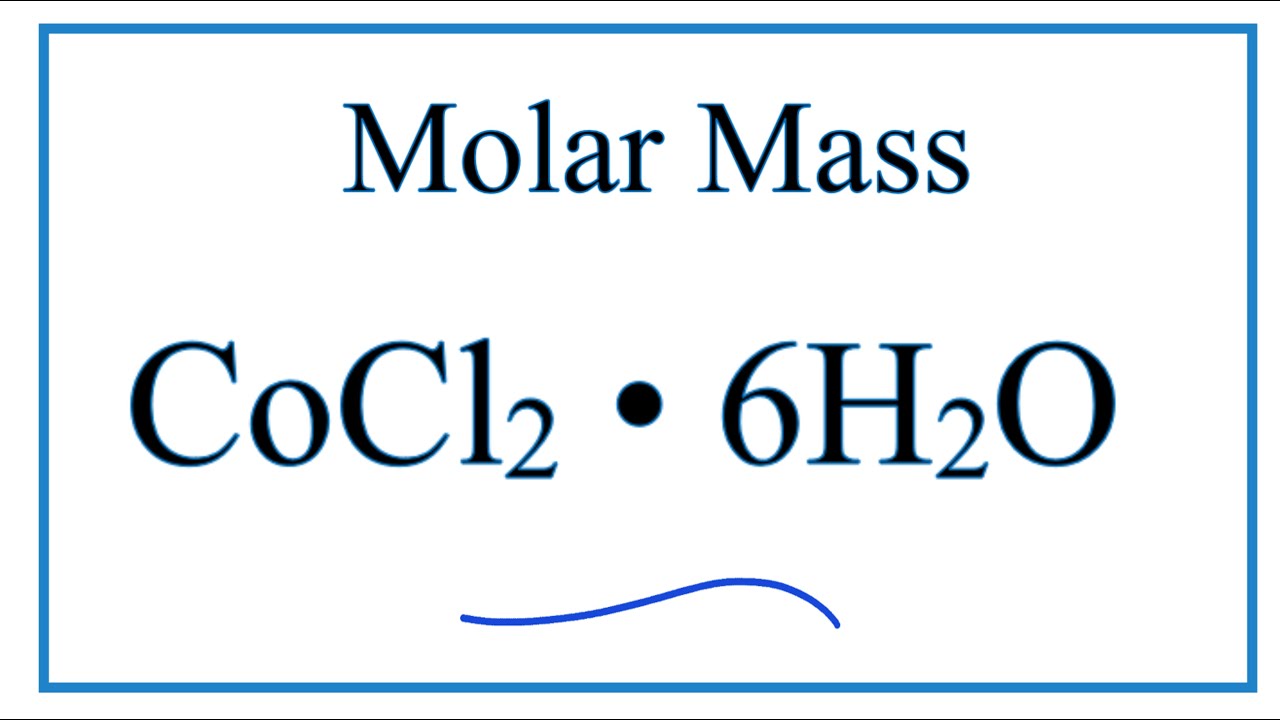
CaCl2.6H2O(s) CaCl2(s) + 6H2O(g); Kp = 6.4 × 10^-17 . Excess solid CaCl2. 6H2O and CaCl2 are taken in a container containing some water vapours at a pressure of 1.14 torr at a

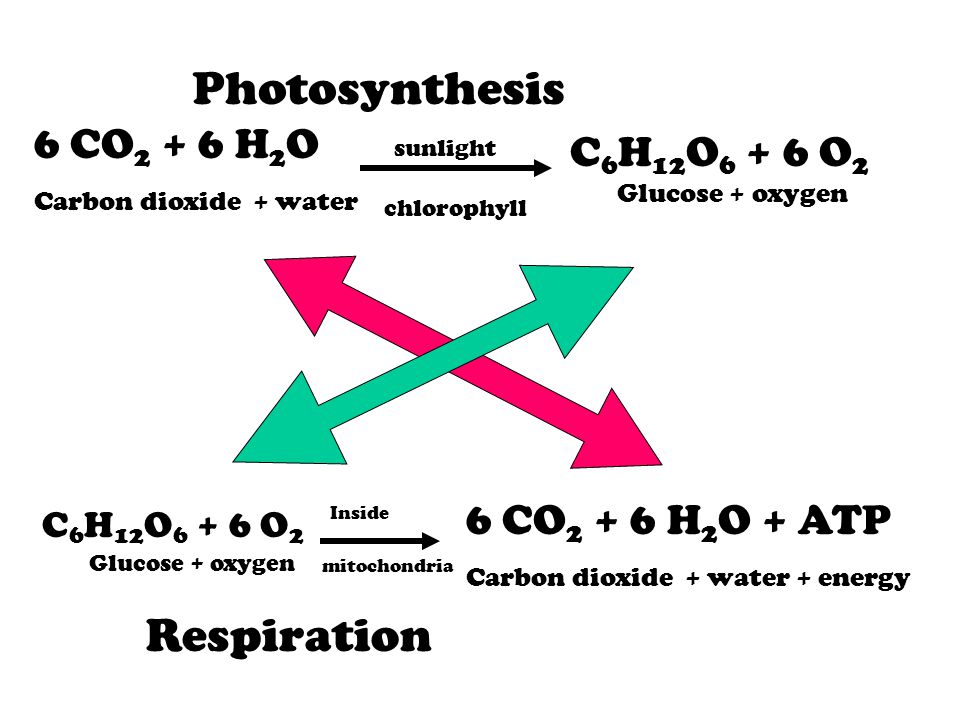
Photosynthesis chemical reaction (Remember… conservation of matter and energy) Light + 6 CO2 + 6 H2O C6H12O6 + 6 O2 + Heat **Remember to look for the. - ppt download
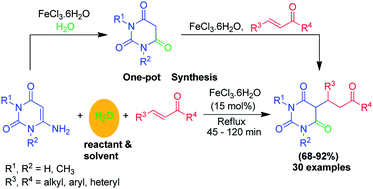
FeCl3·6H2O catalyzed aqueous media domino synthesis of 5-monoalkylbarbiturates: water as both reactant and solvent - RSC Advances (RSC Publishing)

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram
![SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [ SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [](https://cdn.numerade.com/ask_previews/e7bf0f40-8942-4c62-a0e8-90258b6ac48b_large.jpg)
SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [
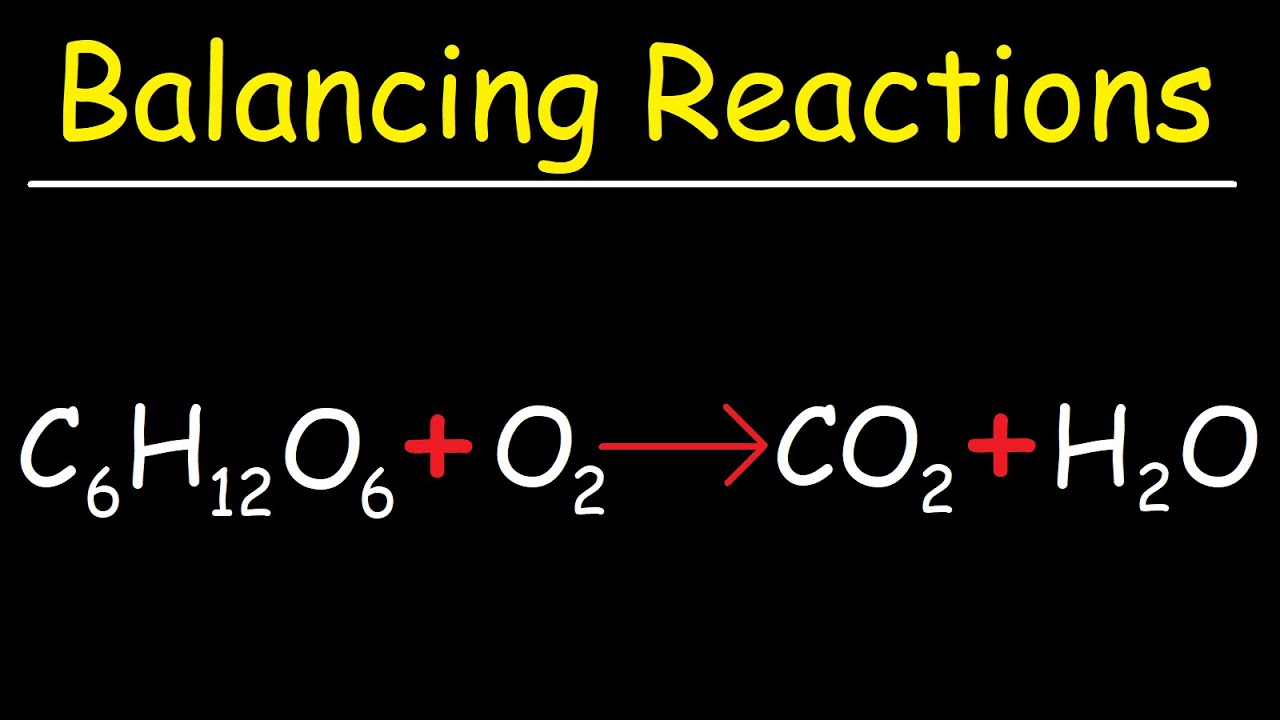
How to balance the Photosynthesis Equation (CO2 + H2O —> C6H12O6 + O2 + H2O? THE BALANCED EQUATION WILL BE OPTION A) 6 CO2 + 9 H2O —> C6H12O6 + 3 H2O + 6 CO2 OPTION B) 6 CO2 + 12 H2O —> C6H12O6 + 6 O2 + 6 H2O
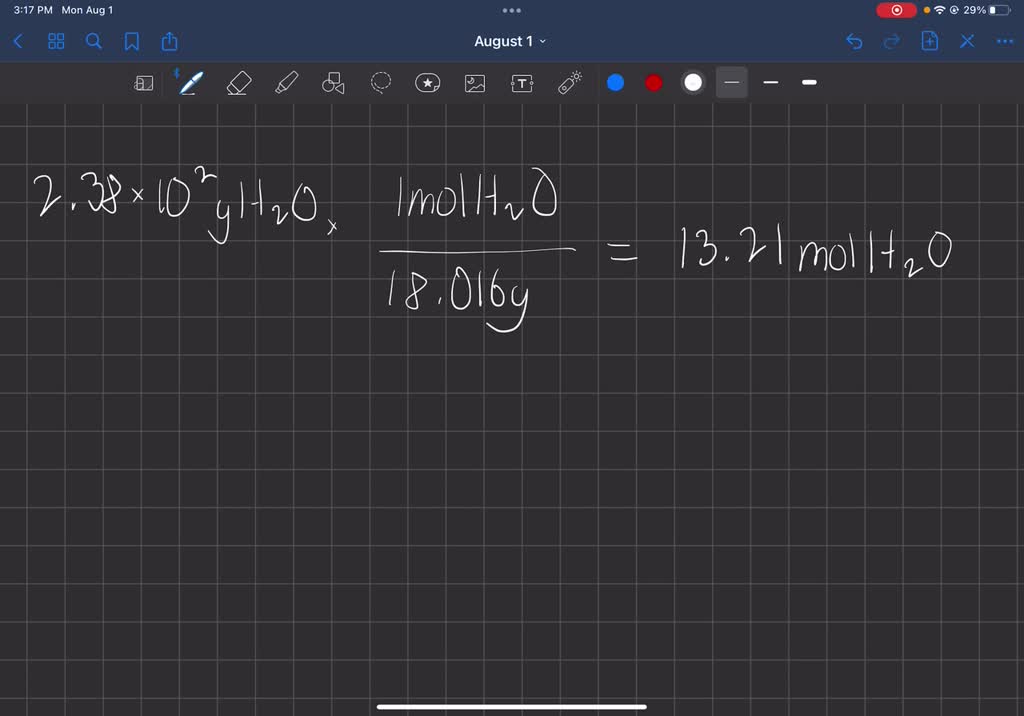
SOLVED: Consider this reaction: 6 CO2 + 6 H2O + light C6H12O6 + 6 O2If there were 2.38 x 102 g of H2O, 18.6 moles of CO2, and plenty of light, what would be the theoretical yield of the reaction?

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram